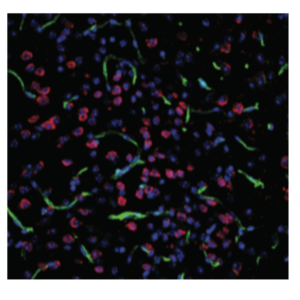
The inability of antibodies and other biologics to penetrate the blood-brain barrier (BBB) is a key limitation to their use in diagnostic, imaging, and therapeutic applications. One promising strategy is to deliver IgGs using a bispecific BBB shuttle, which involves fusing an IgG with a second affinity ligand that engages a cerebrovascular endothelial target and facilitates transport across the BBB. Nearly all prior efforts have focused on the transferrin receptor (TfR-1) as the prototypical endothelial target despite inherent delivery and safety challenges. We have developed bispecific antibody shuttles that engage CD98hc, the heavy chain of the large neutral amino acid transporter (LAT1), and efficiently transport IgGs into the brain parenchyma. Notably, CD98hc shuttles lead to much longer-lived brain retention of IgGs than TfR-1 shuttles while enabling more specific brain targeting due to the apparent lack of CD98hc target engagement in the brain parenchyma. The purpose of this proposal is to address a key challenge related to imaging the delivery and distribution of bispecific antibodies in the brain. We have been using immunofluorescence for imaging the brain, but this method suffers from high background, especially in the aged brain. We propose to develop a mass spectrometry-based method for imaging the brain to address limitations of immunofluorescence that builds on strengths of the three PIs, namely antibody engineering (Tessier), quantitative cellular imaging (Thurber) and mass cytometry (Wen). We are now at the point in which we need advanced methods for imaging the brain, especially the aged brain.
People

Peter
Tessier
ChE
Engineering

Fei
Wen
ChE
Engineering

Greg
Thurber
ChE
Engineering

Colin
Greineder
Emergency Medicine, Pharmacology
Michigan Medicine

Jouha
Min
ChE, MSE
Engineering
Funding

Funding: $45K (2023)
Goal: To develop a mass spectrometry-based imaging method for imaging the brain that overcomes limitations of conventional immunofluorescence methods.
Token Investors: Peter Tessier, Fei Wen, and Greg Thurber
Project ID: 1117
